Understanding the 392 Battery: Technical Features, Equivalents, and Engineering Applications
The 392 battery is a miniature silver-oxide button cell widely used in compact electronic devices that require stable voltage and long operational life. Unlike alkaline coin cells, silver oxide batteries maintain a flat discharge curve, making them ideal for precision electronics such as wristwatches, medical instruments, and measuring devices.
This article provides a technical analysis of the 392 battery, including its chemical structure, electrical characteristics, dimensional standards, and engineering applications. It also examines equivalent battery models and compares the 392 with other similar button cells such as LR41, 377, and L736 to help engineers and device designers select the appropriate power source.
Table of Contents
- 1. What Is a 392 Battery
- 2. Chemical Composition and Electrochemical Reaction
- 3. Key Electrical and Mechanical Features
- 4. Physical Dimensions and Identification
- 5. Engineering Applications
- 6. Equivalent Battery Models
- 7. 392 vs LR41 vs 377 vs L736 Comparison
- 8. Safe Battery Replacement Guidelines
- 9. Proper Disposal and Recycling
- 10. Frequently Asked Questions (FAQ)
- 11. Conclusion
1. What Is a 392 Battery

The 392 battery is a silver oxide button cell designed for compact electronics requiring stable voltage and long-term reliability.
Typical specifications include:
- Battery chemistry: Silver oxide (Ag₂O / Zn)
- Nominal voltage: 1.55 V
- Diameter: ~7.9 mm
- Height: ~3.6 mm
- Typical capacity: 40–45 mAh
Button batteries in the 392 category are commonly used where consistent voltage output is critical, such as precision timing circuits or medical sensors. Compared with alkaline coin cells, silver oxide batteries exhibit lower internal resistance and superior discharge stability, making them more suitable for sensitive electronics.
Because of standardized dimensions, the 392 battery is often labeled using alternative codes such as SR41 or SR736, depending on the manufacturer.
2. Chemical Composition and Electrochemical Reaction
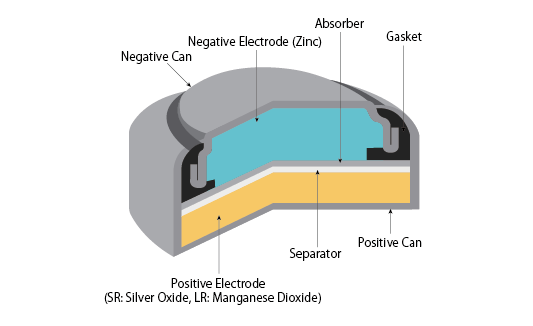
The electrical energy produced by a 392 battery originates from the electrochemical reaction between silver oxide (Ag₂O) and zinc (Zn).
Electrode Materials
| Component | Material | Function |
|---|---|---|
| Cathode | Silver oxide (Ag₂O) | Positive electrode |
| Anode | Zinc (Zn) | Negative electrode |
| Electrolyte | Potassium hydroxide (KOH) | Ion conduction medium |
Electrochemical Reaction
During battery discharge, the silver oxide cathode reacts with the zinc anode to produce electrical energy.
Chemical reaction equation:
Ag₂O + Zn → 2Ag + ZnO
This electrochemical reaction releases electrons through the external circuit, generating electrical power for the connected device.
Engineering Advantages of Silver Oxide Chemistry
Compared with alkaline button cells, silver oxide batteries offer:
- Flatter discharge curve
- Higher energy density
- Lower internal resistance
- Better voltage stability
These characteristics are particularly important for precision electronics, where voltage fluctuations could affect performance or measurement accuracy.
3. Key Electrical and Mechanical Features
The 392 battery offers several technical advantages that make it suitable for low-power electronics.
Stable Output Voltage
Silver oxide chemistry provides a nearly constant voltage of 1.55 V for most of the discharge cycle. This prevents performance degradation in devices such as watches or digital sensors.
High Energy Density
Despite its small volume, the battery can deliver 40–45 mAh, which allows small electronics to operate for extended periods.
Low Internal Resistance
Lower resistance improves load response and enables stable performance even in devices with intermittent current demands.
Compact Form Factor
The small cylindrical form factor enables integration into miniature electronics, including wearable devices and compact sensors.
Leak-Resistant Construction
The sealed stainless steel casing minimizes electrolyte leakage and improves long-term reliability.
4. Physical Dimensions and Identification

Button cell batteries follow standardized dimensions defined by the IEC battery naming system.
Typical 392 battery dimensions:
| Parameter | Value |
|---|---|
| Diameter | 7.9 mm |
| Height | 3.6 mm |
| Voltage | 1.55 V |
| Chemistry | Silver Oxide |
Identification Methods
A 392 battery can usually be identified through:
- Printed battery code (392 or SR41)
- Battery dimensions
- Device manual specifications
- Battery packaging information
Because several button cells share similar sizes, verifying chemistry and voltage is important before replacement.
5. Engineering Applications

The 392 battery is widely used in devices that require low current but highly stable voltage.
Wristwatches
Quartz watches rely on a constant voltage supply to maintain precise oscillator timing.
Medical Devices
Small medical instruments such as digital thermometers and glucose meters use 392 batteries due to their reliability.
Laser Pointers
The battery’s compact size and stable output make it suitable for portable laser modules.
Calculators
Small calculators often rely on button cells for backup or primary power.
Portable Sensors
Miniature measurement devices and sensors also use this battery for low-power operation.
6. Equivalent Battery Models
The 392 battery is sold under several equivalent designations depending on the manufacturer.
Common equivalents include:
- SR41
- SR736
- 392A
- AG3
- LR41 (alkaline alternative)
However, alkaline replacements such as LR41 typically provide shorter service life and less stable voltage.
7. 392 vs LR41 vs 377 vs L736 Comparison
| Specification | 392 (SR41) | LR41 | 377 (SR626SW) | L736 |
|---|---|---|---|---|
| Battery Type | Silver Oxide | Alkaline | Silver Oxide | Alkaline |
| Nominal Voltage | 1.55 V | 1.5 V | 1.55 V | 1.5 V |
| Diameter | 7.9 mm | 7.9 mm | 6.8 mm | 7.9 mm |
| Height | 3.6 mm | 3.6 mm | 2.6 mm | 3.6 mm |
| Capacity | 40–45 mAh | 25–32 mAh | 24–27 mAh | 25–32 mAh |
| Voltage Stability | Very Stable | Moderate | Very Stable | Moderate |
| Typical Applications | Watches, medical devices | Toys, simple electronics | Precision watches | Low-power gadgets |
8. Safe Battery Replacement Guidelines
To ensure device safety, follow these steps when replacing a 392 battery:
- Turn off the device.
- Open the battery compartment using the proper tool.
- Carefully remove the old battery.
- Check the polarity markings (+ and −).
- Insert the new battery in the correct orientation.
- Close the compartment securely and test the device.
9. Proper Disposal and Recycling
Button batteries contain metals that should not enter regular waste streams.
Recommended disposal procedure:
- Store used batteries in a dry insulated container
- Cover terminals with insulating tape
- Deliver batteries to approved recycling centers
Proper recycling helps recover valuable materials such as silver and zinc while preventing environmental contamination.
10. Frequently Asked Questions (FAQ)
1. Is a 392 battery the same as an LR41?
No. Although they have similar sizes, the 392 is silver oxide, while LR41 is alkaline, resulting in lower capacity and less stable voltage.
2. How long does a 392 battery last?
In low-power devices such as watches, it typically lasts 1–3 years, depending on current consumption.
3. Can I replace a 392 battery with an AG3?
Yes, AG3 is often considered an equivalent model, but performance may vary depending on chemistry.
4. Why do watches prefer silver oxide batteries?
Silver oxide batteries maintain a stable voltage output, which is essential for accurate timekeeping.
5. Are 392 batteries rechargeable?
No. Standard 392 button cells are primary (non-rechargeable) batteries.
11. Conclusion
The 392 silver oxide battery is a reliable miniature power source used in precision electronics. Its stable voltage output, compact dimensions, and relatively high energy density make it ideal for devices such as watches, medical instruments, and small sensors.
Understanding the battery’s electrochemistry, dimensional standards, and equivalent models allows engineers and users to select the appropriate replacement battery while ensuring device reliability and safe operation.
